 
Valence refers to the ability of an atom form bonds. The conditional charge of an atom’s atom is called the oxidation state. The terms “ oxidation degree” and “ valence”may not be the same, but they are numerically almost identical. See Also Best Master's of Applied Statistics Degrees for 2022, Online Included Understanding Culturally Diverse Privacy - Aboriginal and Torres Strait Islander peoples' perspectives - Office of the Victorian Information Commissioner The 50 Highest Paying Careers With Only a Bachelor's Degree for 2022 - Online Bachelor Degrees 15 BEST Keyword Research Tools for SEO Numerical values of valence are indicated with roman numerals (I,II, III, IV, V, VI, VII, VIII). It is determined by the number of electrons of an atom spent to form chemical bonds with another atom. It takes values from 1 to 8 and cannot be equal to 0. Valence is the ability of an atom of a chemical element to form a certain number of chemical bonds with other atoms. The number of neutrons found in sodium(Na), therefore, is 12. We know that the atomic numbers of sodium and atomic mass numbers are 11 and 23 respectively (22.989769). This means that neutron number (n) = atomic mass (A) + atomic number (Z). The difference between the number atoms and the number atomic masses is what determines the number neutrons in an element. This means that the sodium atom contains a total eleven electrons. The number of protons found in sodium is 11 The nucleus is protected by a circular orbit (or shell) that contains electrons equal to protons. The number of protons is called the atomic number. 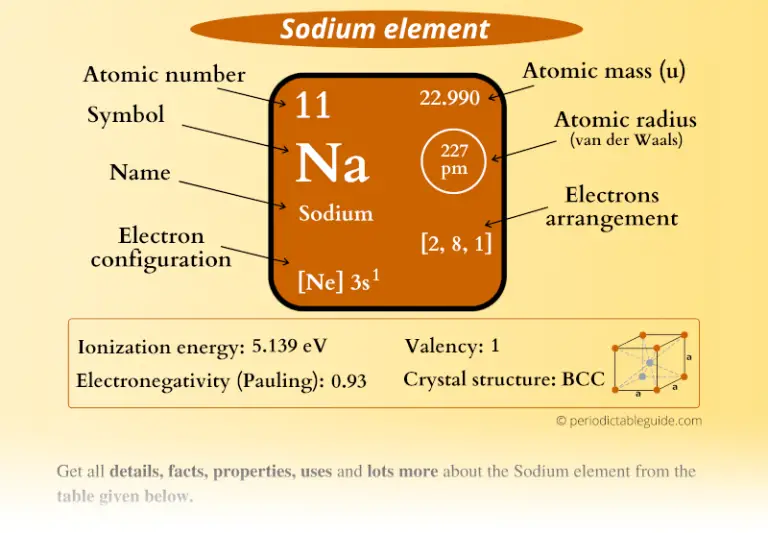
The nucleus is home to protons and neutrons. The nucleus can be found in the middle of an atom. What number of electrons, protons, and neutrons does the sodium (Na) atom contain? This site has an article that explains the electron configuration for sodium(Na).You can read it if necessary. The electron configuration for sodium (Na) indicates that the last shell (orbit) of sodium contains a total electron. They also participate in the formation bonds. The properties of an element are determined by the valence electrons. The valence electrons are the number of electrons remaining in the shell after the electron configuration is complete. The valence electron refers to the number of electrons remaining in the shell’s last orbit. What are the valence electrons for sodium (Na)? It helps cells transmit nerve signals and regulate water level in tissues. A person should only consume 3 grams of salt per day. The human body needs sodium for many functions. High blood pressure can be caused by excess sodium. We have about 100 grams of sodium in our bodies, but we lose it constantly in different ways. atomic numberĪll living things require sodium, something humans have known since prehistoric times. 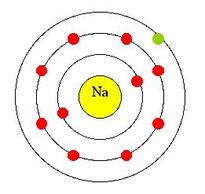
Electrolysis of dry molten sodium chlorineide is used to make sodium metal. It is not found in nature because sodium is so reactive. It can also be found in many minerals, including sodalite, zeolite, and cryolite. However, many salt beds or “lakes” can be found in areas where old seas have evaporated. This salt is very water-soluble and has leaked into the oceans throughout the life of the planet. The most commonly found compound is sodium chloride. The sixth most abundant element on Earth is sodium, which makes up 2.6% Earth’s crust. It can also be used in the chemical industry as a feedstock. It is used in food, and to melt ice on roads during winter. Common salt is sodium chloride, which is the most common sodium compound. A useful sodium salt is sodium carbonate, also known as washing soda. However, sodium salts are more versatile than the metal. Sodium is used in nuclear reactors as a heat exchanger and in the chemical industry as a reagent. Streetlights use sodium vapor to produce a bright yellow light. As a coolant in nuclear reactors, liquid sodium has been used. The production of titan, sodamide and sodium cyanide uses sodium. Because sodium can ignite when it comes in contact with water, it should be kept dry. Sir Humphry Davy was the first to isolate pure sodium by electrolysis of NaOH (caustic soda). While sodium is the sixth most common element on the planet, making up 2.6% of the earth’s crust, it’s a reactive element that is rarely found in nature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
